Pfizer Covid vaccine approved for use in 12 to 15-year-olds
and live on Freeview channel 276
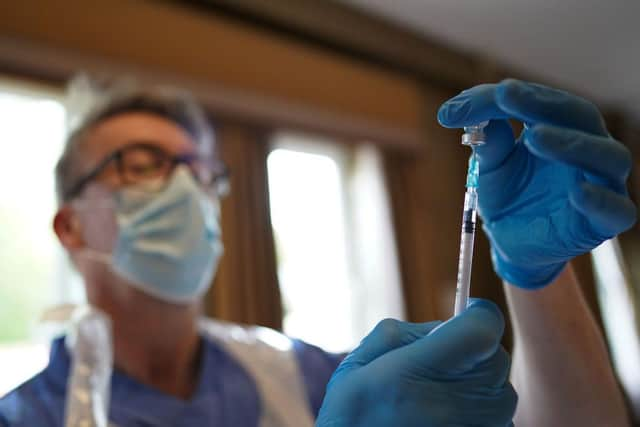
The Pfizer/BioNTech Covid-19 vaccine has been approved for use in children aged between 12 and 15 years old.
The approval from the Medicines and Healthcare products Regulatory Agency (MHRA) comes following clinical trials which determined it is safe to be administered to this age group.
‘Safe and effective’

Advertisement
Hide AdAdvertisement
Hide AdThe trails reviewed the safety, quality and effectiveness of the vaccine among the younger age group, with results finding that the benefits for rolling it out among youngsters outweigh any risk.
The approval comes after the jab was first authorised for use in the UK among all adults at the end of last year.
It is one of four Covid-19 vaccines now authorised to be used in the UK, alongside AstraZeneca, Moderna and Johnson & Johnson, but is the first to be approved for use in children.
Dr June Raine, MHRA chief executive, said: “We have carefully reviewed clinical trial data in children aged 12 to 15 years and have concluded that the Pfizer/BioNTech Covid-19 vaccine is safe and effective in this age group and that the benefits of this vaccine outweigh any risk.
Advertisement
Hide AdAdvertisement
Hide Ad“We have in place a comprehensive safety surveillance strategy for monitoring the safety of all UK-approved Covid-19 vaccines and this surveillance will include the 12- to 15-year age group.
“No extension to an authorisation would be approved unless the expected standards of safety, quality and effectiveness have been met.
When could the jab be rolled out to children?
Dr Raine explained that it will be the responsibility of the Joint Committee on Vaccination and Immunisation (JCVI) to advise on whether children in the 12 to 15 age bracket will be vaccinated as part of the current vaccination programme.
At the moment, people aged between 30 and 39 are the latest age group to be invited to book their jabs and it is hoped that all adults aged 18 and over will be offered their first dose by the end of July.
Advertisement
Hide AdAdvertisement
Hide AdIt has not yet been confirmed if children will now be included as part of the programme.
“The government has asked the independent experts at the Joint Committee on Vaccination and Immunisation (JCVI) to advise whether routine vaccination should be offered to younger people aged 12 to 17.
“We will be guided by the expert advisors and will update in due course.”
We want to hear from you: let us know what you think about this story and be part of the debate in our comments section below
A message from the editor:
Advertisement
Hide AdAdvertisement
Hide AdThank you for reading. NationalWorld is a new national news brand, produced by a team of journalists, editors, video producers and designers who live and work across the UK. Find out more about who’s who in the team, and our editorial values. We want to start a community among our readers, so please follow us on Facebook, Twitter and Instagram, and keep the conversation going.
Comment Guidelines
National World encourages reader discussion on our stories. User feedback, insights and back-and-forth exchanges add a rich layer of context to reporting. Please review our Community Guidelines before commenting.